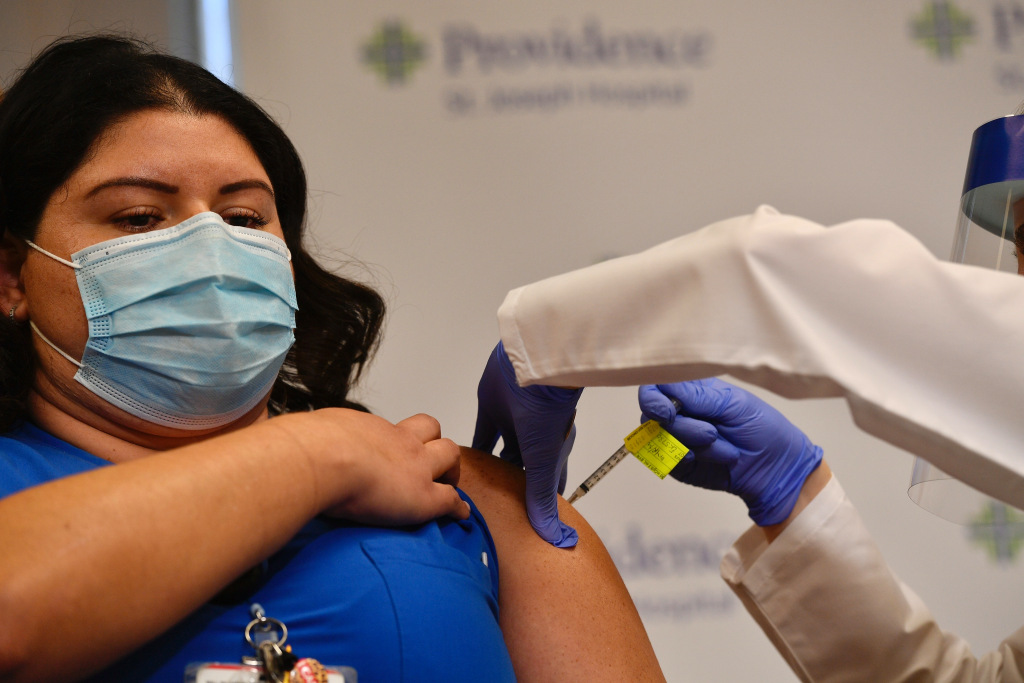
A million vaccinations in the next 10 days.
This is the working goal of one of California’s COVID-19 vaccination planning groups, which convened on Wednesday January 6th and reiterated Governor Gavin Newsom’s request earlier this week to accelerate the state’s mass vaccination campaign.
California is currently in the first phase – Phase 1A – of the campaign, which saw an initial wave of gunfire sent to hospitals and pharmacies to vaccinate frontline health care workers and long-term care residents.
Phase 1A started in mid-December, but it was slow as healthcare providers face the logistical dilemma of hiring new employees to inject other employees in the middle of a winter hospitals rush. There are also healthcare workers who opt out.
Of the roughly 2 million Pfizer BioNTech and Moderna doses shipped to California to date, roughly 490,000 had been administered as of Tuesday, Dr. Erica Pan, California Department of Health epidemiologist and co-chair of the Community Vaccine Advisory Committee, works with the Guidelines for Development Working Group to set the state agenda for coronavirus vaccination.
“We vaccinated a lot of people … but we need to move faster,” said Pan. “Our goal is to manage an additional million in the next 10 days.”
To speed up the first stage of the race, dentists and other medical professionals will be added to the group of personnel authorized to record the shots. Local health authorities have started planning large-scale vaccination sites modeled on coronavirus tests.
It remained unclear when public health officials would advise proceeding to the next stage, Phase 1B, which is divided into two tiers.
Tier 1 comes first and includes people aged 75 and over, as well as workers at high risk of exposure such as teachers and workers in childcare, emergency services, and food and agriculture.
Second is Tier 2, which includes people 65 and over and workers in transportation and logistics, “critical” manufacturing and other sectors, as well as prisoners and the homeless.
Phase 1C would follow and so far comprises people aged 50 and over and people up to 49 years of age with underlying illnesses or a disability that could lead to a more severe case of COVID-19. No vaccine has yet been approved for children under the age of 16.
During the committee’s sixth virtual round table on Wednesday, discussions ranged from distributing the shot fairly to communities at risk of pandemics to announcing ideas for the public sector to increase vaccine acceptance.
“Vaccines don’t save lives, vaccines save lives, so we have to get them out of the fridge – freezer – and into these arms,” said Dr. Oliver Brooks, co-chair of the “Campaign Development Guidelines” working group.